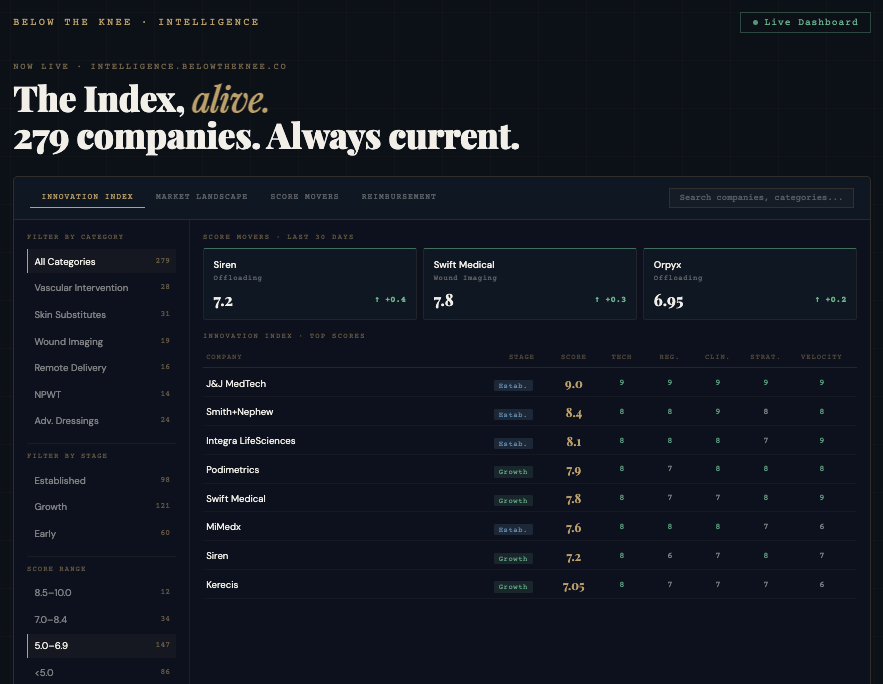
Below the Knee exists to separate signal from noise across wound care, limb salvage, and foot & ankle — tracking the deals, policy shifts, capital flows, and clinical inflection points that actually move this market.
If this sharpened your view, forward it to one person who should be in the room. The best growth in this industry still travels by referral.
March Confirmed the Split
If February set the table, March served the meal — and the menu was not the same for everyone.
Earnings season delivered its verdict on the CMS restructuring in real numbers. Convatec put the largest figure yet on the CAMPs revenue hit: a $30M InnovaMatrix decline, driven by an ~80% CMS price cut. BioStem reported a 55% Q4 revenue collapse as the physician office channel evaporated. On the other side, Sanara MedTech crossed $100M in revenue for the first time, TELA Bio posted record quarterly results after a deliberate commercial rebuild, and InfuSystem's wound care revenue grew over 160% in Q4. The divergence is no longer a thesis. It's a reported outcome.
The regulatory environment bifurcated in ways that will define competitive positioning for years. The FDA loosened its grip on AI-enabled clinical decision support. CMS held its skin substitute restructuring — and received legal cover from value-based care organizations filing an amicus brief in the ongoing litigation. Swift Medical received FDA 510(k) clearance for SwiftRay spectral imaging. Spectral AI landed a $31.7M BARDA award. SINTX Technologies completed the first human implant of its silicon nitride foot and ankle osteotomy wedge. And Aroa Biosurgery announced that its Symphony RCT for diabetic foot ulcers met its primary endpoint — the most significant clinical read-out in the CAMPs category since the restructuring began.
The Stryker cyberattack on March 11 added a dimension no one anticipated: when a single cloud credential can wipe 200,000 devices across 79 countries, the foot and ankle surgical supply chain has a concentration risk that hospital procurement teams are now actively reckoning with.
The filter is working. The market is sorting. See you in Charlotte is next week.
💲 WoundCare Fund — March Summary
March was a month of consistent underperformance against a choppy macro backdrop — two distinct waves of volatility, with early-month small-cap weakness giving way to stabilization as earnings from well-positioned names anchored sentiment.
Weekly Returns: −4.85% | −4.12% | −0.93% | −0.91% The fund outperformed or matched the S&P 500 in three of four weeks — relative resilience in a risk-off environment.
Strength: TELA Bio (record Q4, 16% full-year growth), Sanara MedTech ($100M milestone, 93% gross margins), InfuSystem (wound care revenue +160% in Q4), Advanced Medical Solutions (record 2025 results, woundcare division returned to +9% growth).
Pressure: Treace Medical (−33.81% in the Feb 26–Mar 5 period following Q4 miss and 2026 guidance reset), Organogenesis (−21.62% on 2026 guidance of $350–420M, ~30% below 2025 actuals), BioStem (55% Q4 revenue decline, physician office channel collapse), Polynovo (−13.04%, profit-taking following prior strength).
Takeaway: Capital rotated toward names with reimbursement clarity, services revenue, and documented clinical outcomes. The tolerance for ambiguity continues to compress. Evidence, execution, and channel diversification are the only currencies that matter right now.
📊 Earnings Scorecard
CONVATEC (LSE: CTEC) | Largest disclosed CAMPs revenue impact to date InnovaMatrix declined ~$30M YoY to $69M — the result of an ~80% CMS price reduction effective January 1. Management cited "significant market uncertainty" throughout the call. Critically, Convatec's diversified portfolio (infusion +12.5%, continence +6.6%) absorbed the hit. Pure-play skin substitute companies have no such cushion. This is the data point that frames the stakes for every other CTP player.
BIOSTEM (OTC: BSEM) | Physician office channel collapsed; hospital is the 2026 bet Q4 revenue: $10.1M (−55% YoY). Full-year 2025: $47.5M vs. $69.7M. Q1 2026 guidance: $5–6M. The January acquisition of BioTissue Holdings' surgical and wound assets added ~20 direct reps to support the hospital channel pivot. BioStem is the clearest case study in how the CMS restructuring bifurcated the market by site of care. That pivot is expensive, and Q1 guidance shows exactly how much runway the transition requires.
SANARA MEDTECH (SMTI) | $100M milestone. Clean execution. Q4: $27.5M (+5%). Full-year: $103.1M (+19%), first time crossing $100M. Gross margin: 93%. 2026 guidance reaffirmed at $116–121M. BIASURGE received a Vizient Innovative Technology contract — hospital formulary access at that scale is a distribution moat. Sanara built around the clinical evidence standard the current reimbursement environment rewards.
TELA BIO (TELA) | Record Q4. Commercial rebuild complete. Q4: $20.9M (+18%). Full-year: $80.3M (+16%). OviTex PRS +20% for the year; LiquiFix revenue more than tripled. 89 revenue-generating reps in the field. 2026 guidance: at least 8% growth. ECHO hiatal hernia trial enrolled its first patients — a different surgeon call point from TELA's traditional base, and worth watching.
HUMACYTE (HUMA) | Revenue miss; pipeline milestones intact Q4 revenue: $0.5M vs. $1.35M estimate (63% miss). Full-year 2025: $2.0M from 61 Symvess units. Adjusted EPS of −$0.13 met estimates. Cash: $50.5M. Phase 3 hemodialysis (V012) top-line interim data expected by early June 2026; BLA filing targeted H2 2026. BTIG cut price target to $3 from $6 (Buy maintained); TD Cowen lowered to $1. The June hemodialysis data is the next meaningful catalyst — the platform remains differentiated, but the commercial ramp is encountering real friction.
MEDIWOUND (MDWD) | Contract timing masked real operational progress FY2025 revenue: $17.0M (down from $20.2M) — driven by U.S. government shutdown delaying BARDA contract timing, not operational weakness. Cash: $53.6M (up from $43.6M). EscharEx Phase III VALUE trial on track; Phase II in DFUs and IIT in pressure ulcers launching H2 2026. B. Braun joined the clinical consortium, bringing strategic partners to six — each a potential distribution pathway that doesn't yet show up in the income statement.
📢 Market, M&A & Company Signals
Spectral AI lands $31.7M BARDA award. Additional non-dilutive funding on top of $54.9M already committed, under a contract valued at up to $150M. DeepView is being developed as a triage tool for burn mass casualties — a market where only 250 burn surgeons exist in the U.S. for 40,000 annual hospitalizations. This is strategic government investment in AI wound assessment, not a research grant.
Swift Medical receives FDA 510(k) clearance for SwiftRay. A pocket-sized spectral imaging device capturing bacterial fluorescence and thermal imaging at point of care, regardless of skin tone or lighting — integrated directly into Swift's documentation platform. Bacterial fluorescence makes subclinical infection visible before it becomes clinical. The path from clearance to clinical deployment just got shorter.
Aroa Biosurgery Symphony RCT meets primary endpoint. The Auckland-based company completed its multi-center RCT evaluating Symphony in non-healing DFUs, with preliminary read-out indicating the primary endpoint was met. Why It Matters: This is a preliminary result, not a published study — full analysis and peer review are months away. But Aroa's strategic posture is the signal: they completed this RCT before it was required, betting that the "direction of travel" on evidence standards would make it essential. In a category where the average Innovation Score is 5.93, an RCT that delivers gives Aroa a reimbursement and formulary conversation that most competitors can't have.
Tiger BioSciences launches carePAC™. Pre-configured wound care convenience kits for at-home dressing changes — pre-selected components in a single patient-friendly format. This is a site-of-care strategy as much as a product launch. As post-acute wound care decentralizes, patient-level compliance infrastructure becomes a competitive differentiator.
Mount Sinai deploys OpenEvidence across Epic enterprise-wide. The collaboration makes OpenEvidence accessible to every physician, nurse, and pharmacist across Mount Sinai's seven hospitals and 400+ outpatient practices directly within the EMR. The first enterprise-scale deployment to extend across the full clinical care team. See Special Feature below.
Medtronic acquires Scientia Vascular for $550M. Medtronic's second acquisition of 2026 (following CathWorks at up to $585M) — both are access and workflow plays in vascular. A leaner, vascular-focused Medtronic is a different competitive variable than the sprawling conglomerate of five years ago.
SINTX Technologies completes first human implant of silicon nitride osteotomy wedge. On March 13, SINTX completed the first human surgical procedure using its FDA-cleared SINAPTIC Foot & Ankle Osteotomy Wedge System. Silicon nitride has a two-decade research pedigree in spine; this is its formal entry into foot and ankle reconstruction. Ryan Elmore (15 years in advanced biomaterials) was appointed President concurrently.
Stryker cyberattack wipes 200,000 devices across 79 countries. On March 11, the Iran-linked group Handala gained access to Microsoft Intune and issued a remote wipe across Stryker's global device fleet. 5,000+ Ireland employees were sent home; hospitals could not place Stryker surgical supply orders during the outage. The BTK read: This is not a cybersecurity story. It is a concentration risk story. When a single credential can take down the entire global supply chain of a dominant orthopedic supplier, every OR coordinator and practice manager in foot and ankle surgery needs a backup plan — and a contract that requires one.
Additional signals: Healogics appointed John Landino as CDO (growth-oriented hire with acute care BD background across 600+ wound care centers). FDA issued MediHoney recall for packaging failures affecting sterile barriers — near-term opportunity for competing calcium alginate manufacturers. Advanced Medical Solutions reported record 2025 results with woundcare division returning to growth. Kane Biotech secured VA/DOD distribution for revyve Antimicrobial Wound Gel via SDVOSB partnership.
⚠️ Regulatory & Risk Watch
CMS holds — and gets legal backing. Arnall Golden Gregory filed an amicus brief in CAMPs Initiative v. HHS on behalf of value-based care organizations, arguing CMS acted within its authority and that the prior reimbursement structure drove escalating Medicare expenditures. This is the first significant third-party legal support for the CMS position. Companies waiting on a full rollback need to build their alternative scenario now.
FDA loosens AI CDS classification. January 2026 guidance from FDA Commissioner Makary updated the Clinical Decision Support Software framework — software providing a single, clinician-reviewable recommendation no longer automatically qualifies as a regulated device. For wound imaging, infection detection, and vascular diagnostic tools in development, the regulatory lane just got materially wider. Spectral AI, Swift Medical, NanoBioFab, and RecovryAI (FDA Breakthrough Device Designation) are all better positioned today than twelve months ago.
CAMPs Summit delivered. The March 6–7 West Palm Beach event convened 350+ attendees including KOLs, payer representatives, and commercial leadership from the major CAMP manufacturers. The room consensus: the restructuring is not going away, and Q1 is the trough — not the floor.
🔬 Special Feature — The Regulatory Bifurcation: Two Policy Stories Running in Opposite Directions
The most important strategic dynamic in wound care right now is not a single policy change. It's two policy changes running simultaneously in opposite directions — and the companies that can read both signals at once are the ones that will define the next competitive cycle.
Story One: CMS is tightening.
The $127/cm² price cap, WISeR prior authorization requirements, and LCD withdrawal are functioning as a market filter. Convatec's $30M revenue hit, BioStem's 55% decline, and Organogenesis's 30%+ guidance reset are the documented outcomes. The CAMPs amicus brief filing confirms the legal coalition backing the restructuring is broader than the industry assumed. The filter is working as designed — and it is not finished.
The companies surviving this environment share a consistent profile: RCT or BLA-level clinical evidence, mature reimbursement documentation infrastructure, and channel strategies that don't depend on a single site of care. Aroa's Symphony RCT completion is a direct response to this trajectory. So is Organogenesis's continued investment in Apligraf and PuraPly AM. So is Sanara's Vizient contract. Evidence-first was always the right posture. The CMS restructuring just made it mandatory.
Story Two: FDA is loosening.
The January 2026 CDS guidance update is the most significant digital health regulatory shift in years. Software that supports — but doesn't replace — clinical judgment now has a wider non-device safe harbor. For wound imaging, infection detection, and AI-assisted documentation, the path to market got materially shorter. Spectral AI's BARDA award, Swift Medical's SwiftRay clearance, and RecovryAI's Breakthrough Device Designation all land in a more favorable regulatory environment than they would have twelve months ago.
The Convergence
These two stories aren't competing. They're the same strategy — stated twice, from different agencies.
CMS is demanding that wound care products prove clinical value before accessing reimbursement. The FDA is creating a faster, clearer path for software tools that support the documentation and clinical decision-making that proves that value. Build AI-enabled tools under the new FDA framework. Demonstrate clinical and economic outcomes that justify reimbursement under the tighter CMS standard. That's not two strategies. That's one.
The companies positioned at that intersection — AI-enabled workflow platforms with outcome data and reimbursement clarity — are the ones that will compound in this environment. The ones still choosing between clinical evidence and technology innovation are answering the wrong question.
SAWC will be the first gathering where both stories are in the room at the same time. The sessions on AI documentation, the hallway conversations on Q1 CAMPs utilization, and the WCCC Driving Innovation Summit on April 10 — which puts FDA and CMS leadership alongside industry — will collectively produce the clearest market signal since January 1.
📅 Upcoming Events
April 8–12 | Charlotte, NC 🔥 SAWC Spring / Wound Healing Society Annual Meeting — The world's leading wound care conference, carrying more weight in 2026 than any prior edition. WCCC Driving Innovation Summit on April 10 puts FDA and CMS in the room alongside industry. I'll be at the Swift Medical booth — come find me.
April 8–11 | Washington, DC Diabetic Limb Salvage (DLS) / Wound Healing Foundation — MedStar Georgetown — Premier multidisciplinary limb preservation event running concurrently with SAWC. Essential for anyone tracking PAD, CLI, and DFU outcomes.
May 6–8 | Bremen, Germany 🔥 EWMA-DEWU 2026 — 36th European Wound Management Association Congress. 5,000+ attendees, 150+ exhibitors. The must-attend European wound care event of 2026.
May 14–15 | Austin, TX 🔥 Advanced Wound Care Summit (AWCS) — The business of wound care, not clinical education. Investors, strategics, payors, distributors, and regulators in one room. If you're in commercial or BD, this is May's priority.
September 10–11 | New Orleans, LA 🔥 IPAWS & Tissue Repair Summit (Kernexus) — The Ritz-Carlton — Three tracks: tissue repair, post-acute and mobile wound care, CAMPs market dynamics. The post-acute community's primary gathering — a distinct audience from the hospital-centric SAWC crowd.
October 22–24 | Anaheim, CA DFCon 2026 — ALPS's global interdisciplinary diabetic foot conference. 470 attendees from 27 countries in 2025. First-look venue for next-generation endovascular and AI diagnostic platforms.
December 9–12 | Phoenix, AZ Desert Foot Multi-Disciplinary Limb Salvage & Wound Care Conference — The flagship annual limb salvage event. Federal, VA, DOD, private sector, and podiatric residency communities. Strong hands-on workshop format.
🚀 Looking Ahead
March produced three conclusions that should anchor your operating thesis through SAWC and into Q1 earnings.
The CAMPs market is contracting before it matures — and the evidence bar is the dividing line. Convatec, BioStem, and Organogenesis documented what happens without it. Sanara, TELA Bio, and Aroa documented what happens with it. The amicus brief makes clear the legal environment isn't reversing. If your 2026 commercial plan still assumes a rollback, build the alternative now.
Site-of-care migration is the infrastructure story no one is moving fast enough on. McKesson's distribution expansion, Tiger's carePAC™, SAWC's Mobile Wound Management Live series, and BioStem's full channel pivot are all pointing at the same shift. The physician office channel for CAMPs is contracting. Home health, post-acute, and outpatient are absorbing volume — with different economics, different compliance requirements, and different channel infrastructure. The companies and distributors positioning now will capture disproportionate share when the transition stabilizes.
AI integration is moving from tool to infrastructure, and the window to position correctly is narrow. SwiftRay clearance, Spectral AI's BARDA award, RecovryAI's Breakthrough Device Designation, and Mount Sinai's enterprise-wide EMR deployment are different layers of the same emerging stack. Wound care vendors that don't treat EMR interoperability as a commercial strategy — not just a product feature — are building toward a distribution disadvantage. The Mount Sinai announcement set a new institutional baseline. SAWC will tell us how many wound care companies have registered what that means.
Q1 2026 earnings will begin in late April. SAWC will deliver the room-level read that no earnings call can. The opportunity is real. So is the work required to capture it.
Let's keep building.
— Scott
2026 Market Report
If these insights resonate and you're navigating growth, portfolio strategy, or commercialization in wound care or limb salvage, I work with teams facing exactly these decisions. → belowtheknee.co/consulting
Below the Knee is an independent market intelligence newsletter covering wound care, foot & ankle, and limb salvage markets. Analysis and opinions are solely those of the author and do not constitute investment advice.